Alfred Sommer is dean emeritus at the Johns Hopkins Bloomberg School of Public Health. He is known for demonstrating that vitamin A supplementation prevented infections and blindness, an achievement for which he won the Albert Lasker Clinical Medical Research Award in 1997.
Q: What drew your attention to epidemiology in the context of global health?
A: I was interested in why people get disease, who gets disease, and how we can prevent it. That led me to epidemiology and to global health because the issues seemed so large and unattended. If you go back in history 50–80 years ago, there was a spurt of research activity during the colonial period. After that, very few people had either the interest or the adequate support to systematically study diseases in the developing world, so it was a wide-open area.

In 1997, Alfred Sommer traveled to Kathmandu to work on a large randomized clinical trial that eventually demonstrated that one large dose of vitamin A twice a year reduced child mortality by one-third. Since he arrived on his birthday, the staff met him with an elephant carrying a banner wishing him a happy birthday, which he rode from the airport into town.
Q: What are some of the major economic and scientific reasons to invest in preventing and treating diseases that predominantly affect developing countries?
A: The major reason is self-interest. Except for a few rare diseases in very tropical areas that are not particularly replicated in the West, almost any disease that occurs in the developing world can occur here. These are largely infectious diseases, and the speed at which they spread is as quick as one jet airplane ride. We think, “Well, that is a tropical area; we should not have to worry about it; they have a lot of mosquitoes.” Yet, everybody suddenly woke up to the Zika virus epidemic last year in Brazil, which made its way to Miami, Florida, in very short order. We certainly remember severe acute respiratory syndrome (SARS), which probably began in southeastern China and spread from animals to humans. SARS was a virus that was a very near miss. It was very, very deadly. We were fortunate that it was not terribly contagious, which is the reason that most of the secondary cases were in hospital workers or people who were in close contact with those who had the disease. But if it had the communicability of something like flu, it would have wiped out perhaps hundreds of millions of people, if not more.
Q: Was the relatively low disease impact in our part of the world thanks to good responses, or have we been just lucky?
A: We have mostly been lucky. With SARS, people clamped down and screened the population very heavily in China, Hong Kong, Taiwan, and Canada, where they had major outbreaks. Within a few months, we knew what virus it was, its structure, and its genetics. But we did not have a vaccine. For viruses, we really depend on vaccines, and we do not have vaccines against very many tropical diseases.
Aside from tropical diseases, the next great pandemic could be a simple influenza virus, which we have been dealing with for decades. We have old technology even for influenza. The vaccines have to vary every year because the virus changes every year. We have not yet developed the technology to make a vaccine that attacks a part of the virus that is preserved. Instead, our vaccines target the parts that change every year, and even when we get a perfect match between the vaccine and the viral strains, the flu vaccine is about 65% effective overall. It is probably not effective at all in somebody my age because the ability to develop an immune response declines with age. We still have not cracked the code to make an effective vaccine for influenza like we have been able to make for measles and polio. And we do not have vaccines for the viruses that have yet to emerge and can suddenly make their way to the US, Europe, and other areas that otherwise have good health services.
Q: What other critical elements need to be addressed for effective disease response?
A: Vaccines are probably the single most important area for having the ability to respond to viral outbreaks, but we also need a much stronger early surveillance system; that is where epidemiology comes in. If we are not identifying new and emerging agents, then we are not in a position to make a vaccine for them. It is just as important to have early surveillance of zoonotic diseases—not just human diseases—because often the organisms that attack humans begin by spreading through animals. This is a tough thing to do because most of the places where these diseases emerge have weak health systems and rudimentary surveillance programs.

In 1982, Sommer conducted a course on epidemiology and statistics for the chairmen of ophthalmology in China through the World Health Organization. This photo was taken at the opening ceremony.
In 1982, Sommer conducted a course on epidemiology and statistics for the chairmen of ophthalmology in China through the World Health Organization. This photo was taken at the opening ceremony.
Q: Is improving disease surveillance only a matter of more financial investment, or do we also need to develop long-term relationships with the relevant countries?
It is all of the above. The Centers for Disease Control and Prevention, the World Health Organization, and a number of private philanthropies like the Gates Foundation and the Chan Zuckerberg Initiative have been trying to stimulate and develop the surveillance systems of poor and middle-income countries. But the resources are a fraction of what we need. The commitments on the parts of governments are limited because they have what they see as more pressing needs. That does not even include conflict zones, which have been a challenge.
A good example is the effort to eradicate polio, which is something that we should have accomplished years ago. We got it down to one or two countries that had naturally occurring polio cases, but we have not been able to eliminate the disease entirely because of the conflict zones, particularly Afghanistan and Pakistan. Little outbreaks keep popping up, and we are not allowed to vaccinate the children in those areas. Of course, when polio occurs in one area, it can spread to other areas, so this is a constant battle. The trouble is that the world loses interest and enthusiasm, so we need to finish this job with polio in the next five years or so. That is just one disease; you have all these others waiting in the wings. They have got to be detected early, and we have got to be able to respond to them early. Part of this will certainly be an attempt to make vaccines, but a big part will be finding an outbreak and clamping down on it.
Q: You have referred to epidemiology as “Sherlock Holmes” played out in the medical arena, but it seems that today it is more important to identify a potential epidemic before it happens. In that context, is detective work still as important today as it was 20 years ago?
A: We would like a very early signal that indicates something that we need to contain. But even if you detect that signal, how do you identify whether that signal is real or not? What does that signal mean? Where did it come from? And what is causing it? All of that requires strong, important detective work that is epidemiological in nature.
Today we have a lot more tools that we can use in our detective work than we had 50 or 60 years ago. These include genomics, so you can very quickly identify the gene structure of the organisms causing the problem, and molecular tools to multiply an organism so that you can begin to study it. We have tools that allow us to pick up the signal, such as computational biology, and connectivity in data sampling methods. A method that has been used repeatedly lately is syndromic surveillance, which monitors Google searches and internet activity. Are people suddenly buying more flu or cold medicines or antibiotics than they were on an average basis? Does that mean that something is going on? Also, with the use of GPS, you can precisely locate where things are happening; you can see the houses or the huts and how they are arranged and identify the route by which an illness may be transmitted. This is something we could not do before. I used to have to go trudging through the field to map the houses. All these are terrific new tools that dramatically enhance our ability to conduct detective epidemiological work.
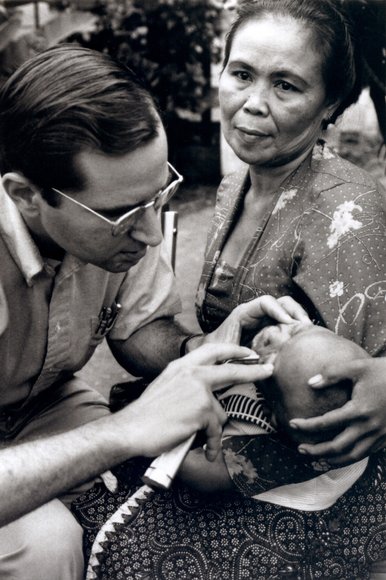
At the Cicendo Eye Hospital in Bandung, Indonesia in 1976, Sommer examined a child suffering from “nightblindness,” the earliest ocular manifestation of vitamin A deficiency.
Q: Once we identify what needs to be done for a particular disease or outbreak in a developing country, what is the path to implementation?
A: It varies with the disease and the risk it raises for countries that have the resources to make a difference. Cholera has been a deadly disease primarily in Asia, although it periodically breaks out of its home territory. When it starts to threaten wealthier countries, then money goes into making a vaccine and developing strategies to contain the outbreak. We have a vaccine now. It is not the best vaccine, but we have one, in part because Haiti got inundated with an epidemic. They are near our back door. For the past 50 years we had no vaccine because the countries that had cholera did not have the scientific and financial resources to develop one, and cholera was not a threat to the countries that did.
Q: In the late 1970s, you worked on the effects of vitamin A deficiency, which seemed less relevant in the developed world. Despite the implications of vitamin A in a wide spectrum of diseases, there was little research interest in that area. You referred to this as a profound amnesia. Are there examples of such amnesia today?
A: The big issue right now is not a scientific one; it is more of a sociobehavioral issue: antivaccine politics in the US and Europe among large communities of well-educated individuals. We have tried everything we can to correct the trend. The problem is that the people who are against vaccines keep moving the goalpost. Doing proper science is critically important, but you can design the perfect study to prove that it does not cause autism and they will say autism was not the issue, or that they are now concerned about a different combination of vaccines. One paper they keep referring to has been debunked and taken out of the literature. The Lancet journal that initially published it has retracted it and said it was all rubbish. Yet it has stirred some primeval fear amongst a large group of parents.
Vaccine avoidance is almost never a problem when someone has a disease and is going to die from it. The problem is preventing a disease in the first place. If prevention is successful, nothing happens, and that is not dramatic. As D. A. Henderson, who ran the smallpox eradication program, would commonly say, nobody is grateful today that they are not going to die of smallpox. It is out of sight, out of mind. If we suddenly had a new smallpox epidemic, or if we had something like the 1918–1919 flu epidemic, people would rush to get vaccinations.
Last year we gave the Lasker Award for the human papillomavirus (HPV) vaccine, which can prevent 95% of HPV infections that cause cervical cancer. Essentially in one generation, no woman would ever need to have a Pap smear done because there would be no more cervical cancer. While not a lot of women die in the US from cervical cancer because we pick it up early, they still need regular Pap smears, biopsies, or surgical procedures if they start to get precancerous lesions. We can make that all go away if we vaccinate all boys and girls in their young teens. Yet there are large segments of the population in the US—including whole states—that will not support that approach to a perfectly preventable disease. They come up with all kinds of reasons such as if teenagers are vaccinated, they are more likely to have sex. Why would they? They are getting a vaccination that will make it less likely for them to get a viral infection that might lead to cervical cancer in females or genital warts and other unpleasant outcomes in males. So we have a real behavioral issue here, which we somehow must figure out how to attack.
Q: What would you tell today’s physician scientists to inspire them to enter the global health field?
A: The young students I see today are motivated and excited about global health. The problem is that they need funding to support their work. Funding agencies have a greater interest in global health these days because they recognize that whatever occurs overseas is not going to stay there, and we have greater empathy for the concerns and needs of people overseas. Still, funding is tighter today than it was when I first became involved in biomedical research. There is no question about that.
An even greater problem is that most medical students now finish medical school with an average of about $180,000 in debt. Just think about that. They come out of medical school still facing residency and fellowship training, and they are already $180,000 in debt. It is not only driving them out of doing things they would really like to do, like getting involved in tackling global health problems, but it is also a big determinant of the subspecialties they choose. The biggest impediment today—and one that gives me great concern—is the indebtedness of people as they finish their medical training. That is what limits their free choice.

In 1986, Hugh Downs and his team from 20/20 decided to cover Sommer’s vitamin A research trial in the Philippines. “I vividly recall him calling me at Hopkins to say they were planning to do this the following week and expected me to be there,” Sommers recalled. “I told them I knew nothing about it, and had a long list of surgical procedures scheduled that week. He replied, ‘If you don’t go, we don’t go.’ I went.”
